Plasma
Adhesion Promotion
With a plasma treatment it is possible to achieve an optimal adhesion between two materials at the whole interface. The adhesion is provided by forming chemical covalent bonds at temperatures below 50 °C. Plasma treatments create either reactive coatings on the surface or chemically functional groups and radicals in the surface. This allows to connect metals chemically with plastics as well as plastics with plastics.


Adhesion of plastics on plastics
Many plastic types are inert against most chemicals under standard conditions. That means that one can even dissolve plastics but the polymer chains do not chemically react. Polyethylene (PE) is for example inert against acids and bases and gluing is only possible with high effort. In comparison polyamide can easily be glued because of the chemically reactive groups in the polymer. These so-called functional groups in the can form chemical bonds to another polymer. The result is an ideal adhesion. The energy in the plasma can crack chemical bonds in the polymer of plastics. The open bonds can react with chemical substances (for example glues) or functional groups can be attached to them in the plasma.
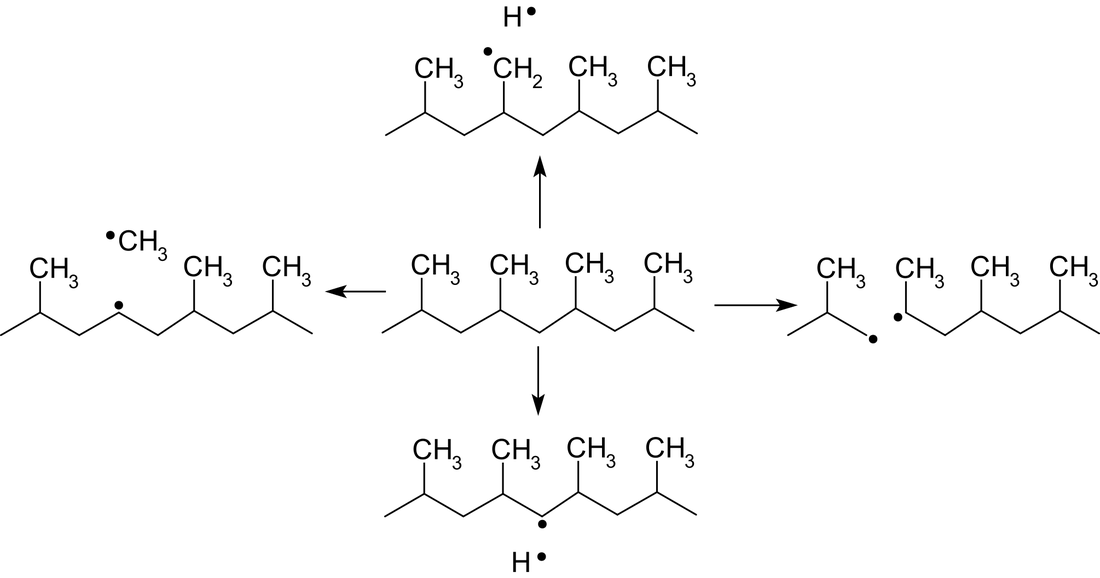
The most simple method to chemically activate plastics is the usage of an argon plasma. The substrate is hereby put into a vacuum chamber that will be filled with the noble gas argon. By applying an electric voltage to an electrode in the chamber some of the argon atoms are ionized and a plasma is ignited. The argon ions try to return into an electrically neutral state by catching an electron. The reactivity of the ions is so strong that electrons are removed from the chemical bonds of the polymers. The result are open bonds (unpaired electrons) in the plastic surface, see Fig. 1.
The electromagnetic radiation created in the plasma is strong enough to crack bonds in the polymer. By excitation of atoms and molecules in the plasma radiation is emitted in the range of infrared to ultraviolet (UV). The UV radiation is the part that has enough energy to crack bonds. The possible reactions for polypropylene (PP) are shown in Fig. 2 as example.
Plasma treatments with argon have the advantage that the surface chemistry of the surface is not changed. The disadvantage is that the open bonds recombine quite quickly. To have open bonds also after some hours after the activation the plasma process time needs be be quite long (several minutes). A side effect is that open bonds of different polymer chains can react with each other. Chained polymers will therefore be cross-linked. This effect can intentionally be used to increase for example the shore hardness at the surface of elastomers.
The part itself keeps its elasticity as before the plasma treatment. The surface however has a lower surface energy so that parts don’t adhere anymore at each other and most particles from the environment don’t stick anymore on the surface. For polymers that strongly react with UV light, like e.g. polyoxymethylene (POM), the plasma treatment can change the bulk properties permanently. Depending on the UV penetration depth the plastic gets cracks or staining. As that change depends on the radiation dose, these materials should be treated as short as possible. The bulk material needs to be tested afterwards.
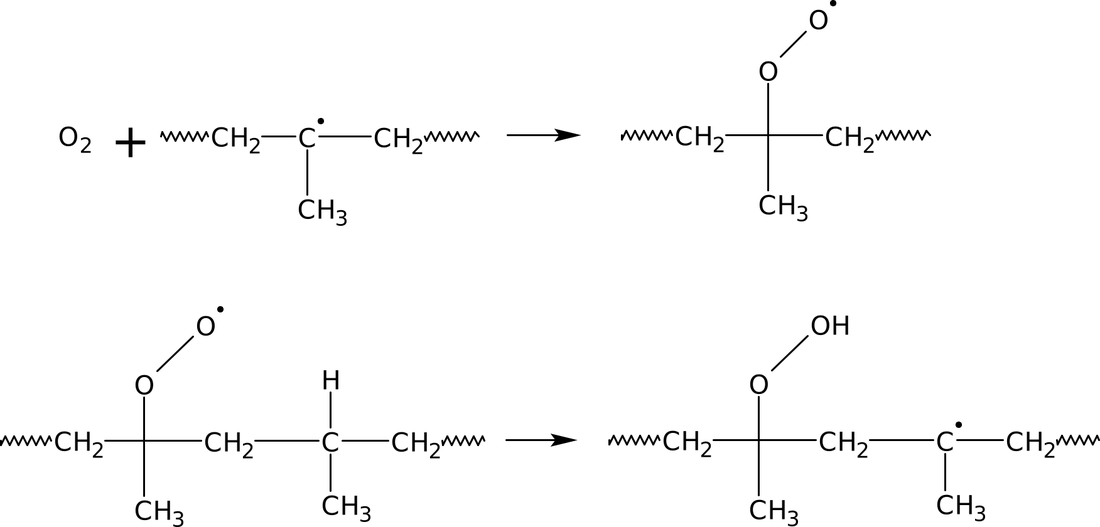
By using a plasma consisting of molecules, the created open bonds can be saturated with functional groups. One of the most used gas for the activation of plastics is oxygen because it creates quickly (within seconds) hydroxyl groups (OH groups) in the surface. Fig. 3 shows the occurring chemical reactions.
If the oxygen plasma treatment time is too long the plastic will be oxidized. The surface is then not only activated but also etched.
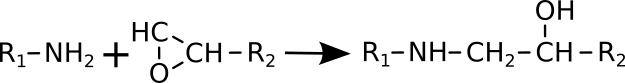
The hydroxyl groups in the surface are able to react with other chemical groups in the surface of a second material resulting in a covalent bond between the two materials. For example OH groups can react with NH2 groups (amino groups) in a condensation reaction by loosing a water molecule.

Possible chemical reactions
Tab. 1: Possible chemical reactions of commonly used functional groups
Hydroxy + Amino

Hydroxy + Epoxy

Hydroxy + Carboxy

Hydroxy + Vinyl

Hydroxy + Isocyanat

Amino + Epoxy
Amino + Carboxy

Amino + Vinyl

Amino + Isocyanat

Vinyl + Thiol

Tab. 1 lists the chemical reactions of functional groups that are often used for adhesion promotion. Due to the various plastic additives it is necessary to test in every individual case what functionalization and adhesion promoter can be used in practice.
For the plasma treatment of plastics it is important to understand that polymer chains are movable. The chains can rotate so that functional groups that were attached to the polymer in a plasma process will not stick out of the surface after a certain time. They are then not available for reactions. A direct activation of chained polymers is therefore temporally not stable. For example, the activation with oxygen lasts for typical PE types only a few hours up to 2 days. Fig. 7 illustrates the effect of the rotation of activated polymer chains.
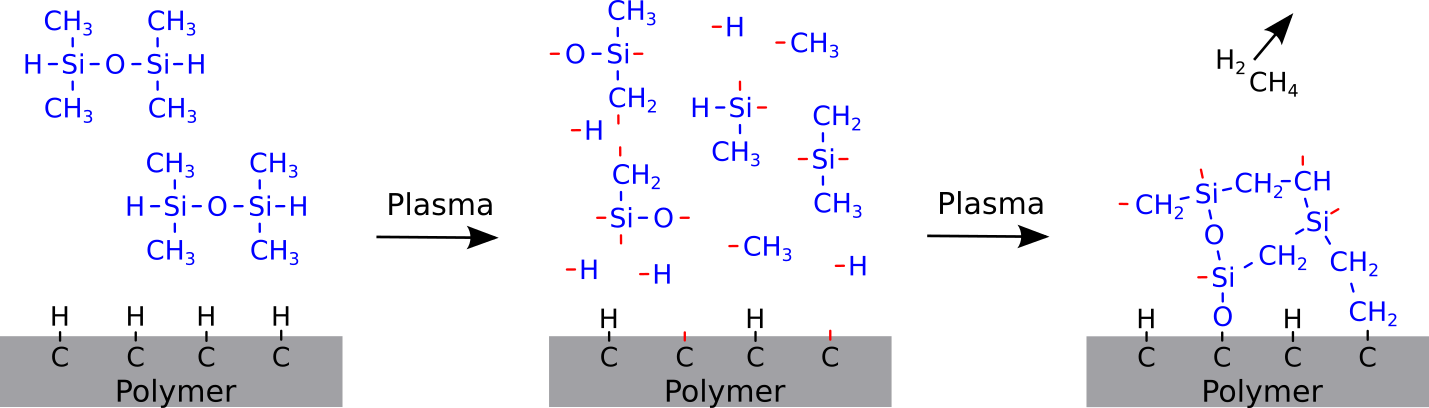
Within cross-linked polymers the mobility of the polymer is very limited because the chain segments are short. In heavily cross-linked polymers (thermosetting polymers) the cross-linking is so strong that an activation is usable up to weeks. This property can also be used for low cross-linked or chained polymers by applying a heavily cross-linked polymer onto their surface. In this case the polymer is at first activated in a plasma. Subsequently a plasma is ignited in a gas consisting of so-called precursor molecules. Fig. 8 shows the principle of the plasma polymerization. The precursor molecules are fragmented and ionized in the plasma. When the fragments and ions hit the activated surface they are chemically bound to it. The molecule fragments form this way a plasma polymer coating. Plasma polymers are heavily cross-linked and don’t contain defined repeat units. Therefore e. g. silicone-based plasma polymers have different properties compared to chained silicones.
In fact, by activating the applied plasma polymer coating one activates a layer of thermosetting polymer and the activation is therefore usable for a long time.
Plasma polymer coatings are not in every case necessary for a temporally stable activation. If an adhesion promoter consisting of large molecules is applied directly after the activation the molecules cannot rotate into the polymer because of their size, see Fig. 9.
All metals (with few exceptions like gold) and metal alloys have a native oxide layer at their surface that also contains hydroxyl groups. The oxide layer can be used for adhesion promotion by applying a plasma polymer onto it. Depending on the material of the substrate it is advantageous to oxidize the surface in an oxygen plasma in advance. The plasma polymer is covalent-bonded to the substrate and the above described techniques for adhesion of plastic on plastic can be used.